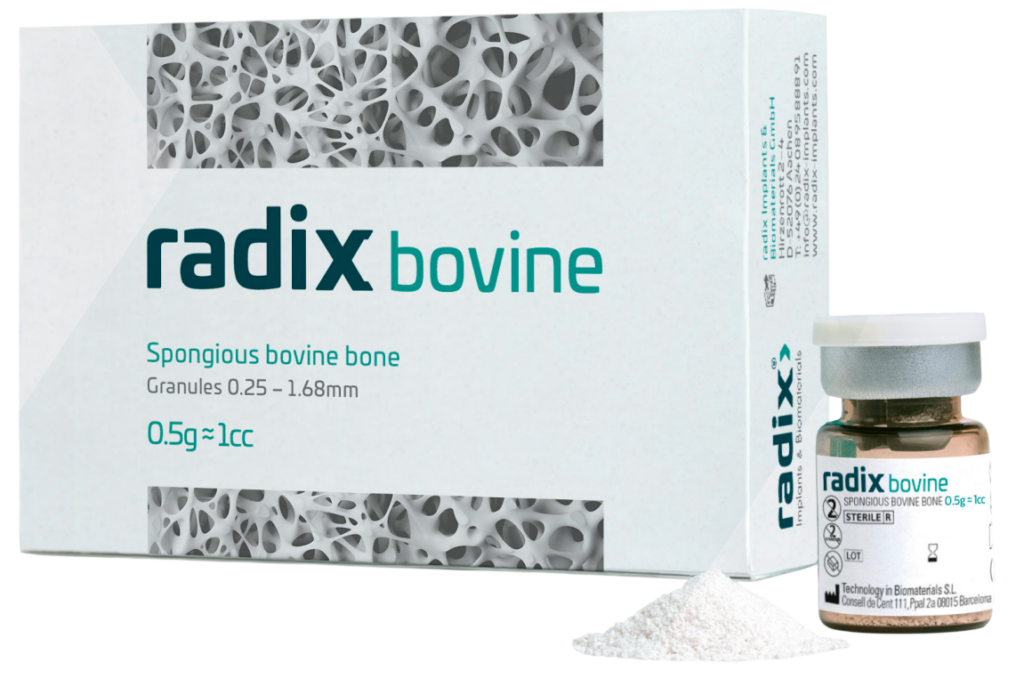
radix bovine – Spongious bovine bone
Due to the characteristics of its natural and inorganic structure, radix bovine may be compared to
human bone. Its interconnected pore structure and special consistency, it promotes bone growth in the
implant zone, and undergoes a gradual physiological remodeling process through the osteoclasts and
osteoblasts. radix bovine is an excellent alternative to autologous bone.
The manufacturing based on different physical-chemical processes removes all organic components
from the femur proximal extremity (femoral bovine head) guaranteeing high safet

![]()
> Bone graft 100% natural
> Easy handling
> Rapid bone regeneration
> Excellent osteoconductivity
> High wettability
> Maximum efficacy and safety in bone regeneration
![]()
> Reconstruction
> Increase of alveolar ridges
> Bone dehiscence
> Filling of immediate implants
> Sinus lift procedures
> Preparing implant sites
> Filling of bone defects
Product specifications:
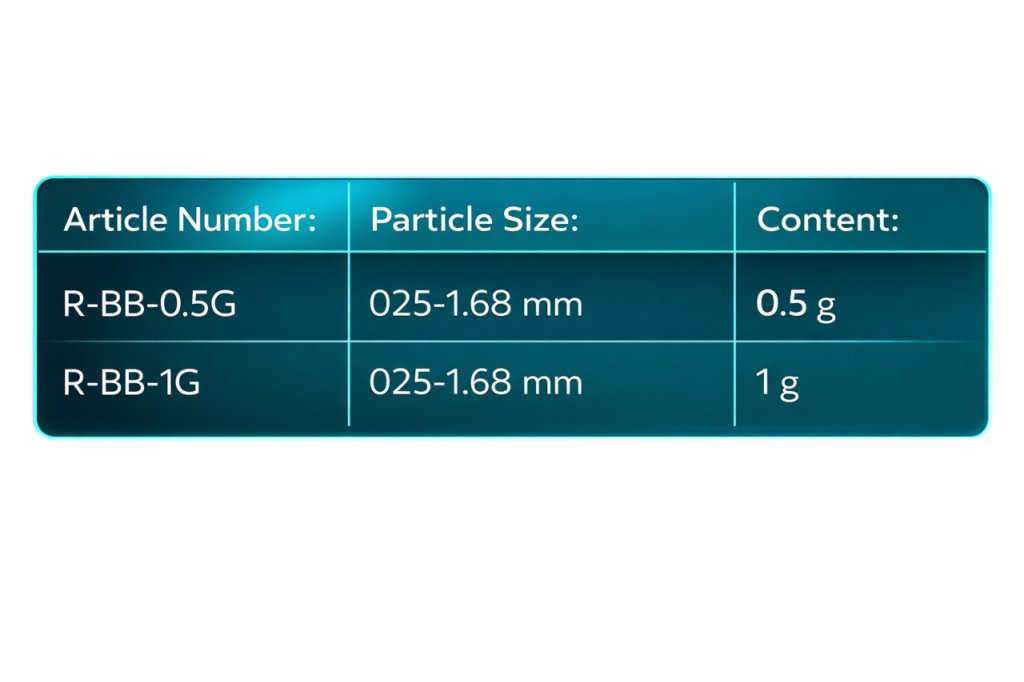
radix bovine Spongious bovine bone is available in granules with a particle size between 0.25-1.68 mm: it is presented in gamma radiation sterile vials of 0.5g and 1.0g.



Manufacturing process:
radix bovine is entirely made of the femoral head of cattle. The origin of raw material and
type tissue used and the manufacturing process of this bovine material meets the safety criteria and requirements.
Therefore, the risk of BSE transmission can be considered negligible.
BSE prevention regulations:
radix bovine is a medical device manufactured utilizing animal tissues and therefore
classified as class III according to the Rule 17 of Annex IX of Medical Device Directive 93/42/EEC. The BSE/TSE safety
concerns have been addressed in accordance with Commission Regulation (EU) No 722/2012.
The characteristics of the natural and anorganic structure of radix bovine can be compared to that of human bone.
